And the Van der Waals radius is always higher than that of the covalent bonding radius. We express atomic size in terms of Van der Waals radius since they do not form covalent bonds. Therefore the inter-electronic is maximum. And the reason is that inert gases have completely filled orbitals. The atomic radius abruptly increases as we get from halogens to inert gases. Variation of Atomic radius Within a Period This measure of atomic radius is known as the van der Waals radius after the weak attractions present in this situation. The attractive forces between the atoms are much less, and the atoms are importantly “unsquashed”. 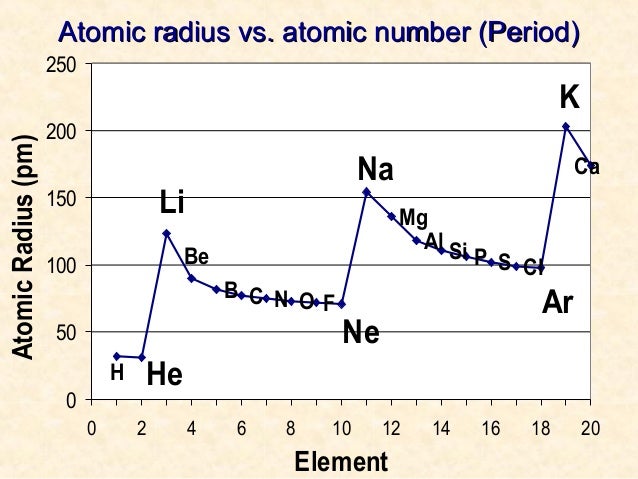
As an example, the internuclear distance between the two hydrogen atoms in an H2 molecule is measured to be 74 pm. The type of atomic radius being measured here is known as the metallic radius or the covalent radius determined by the bonding.The units for measurement of atomic radii is picometer ,which is equal to 10−12 metres. This is what you would get if you had metal atoms in a metallic structure, or atoms that are covalently bonded to each other. The atoms are pulled nearly together and so the measured radius is less than if they are just meeting. Same atoms can be initiated to a different radius depending on what is surrounding it. The radius of an atom can only be initiated by measuring the distance between the nuclei of two connecting atoms, and then by halving that distance. Metallic Radius:- Metallic radius is explained as a half of the distance between the nuclei of two atoms in a crystal or between two nearest metal ions in the metallic network.Īn atom doesn’t have a fixed radius. The Van der Waals radius magnitude is dependent on the configuration of the atoms when the elements are in the solid-state. Van der waals Radius:-It is a half of the distance between the nuclei of two similar non-bonded lonely atoms or two nearest similar atoms belonging to two adjacent molecules of an element in the solid-state. The internuclear distance between two bonded atoms is known as the bond length. Therefore, m covalent is equal to ½ (internuclear distance between the two bonded atoms). TYPES OF ATOMIC RADII There are three types of atomic radii:-Ĭovalent Radius:-Covalent radius is a half of the distance between the nuclei of two covalently bonded atoms of the same element of a molecule. It is also known as the distance measured from the centre of the nucleus to the point until the density of the electron cloud becomes maximum. Atomic radius is the distance measured from the centre of the nucleus to the external shell carrying electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
